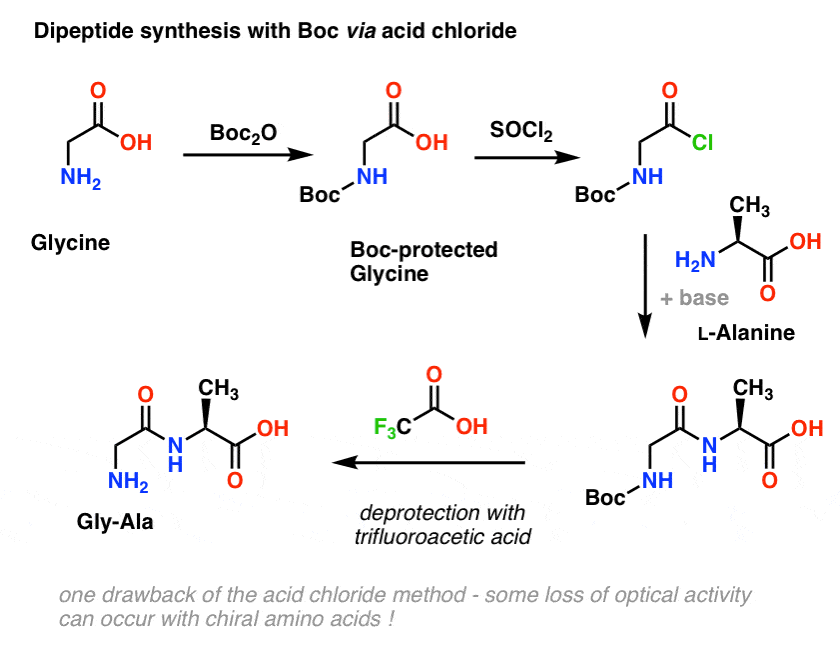
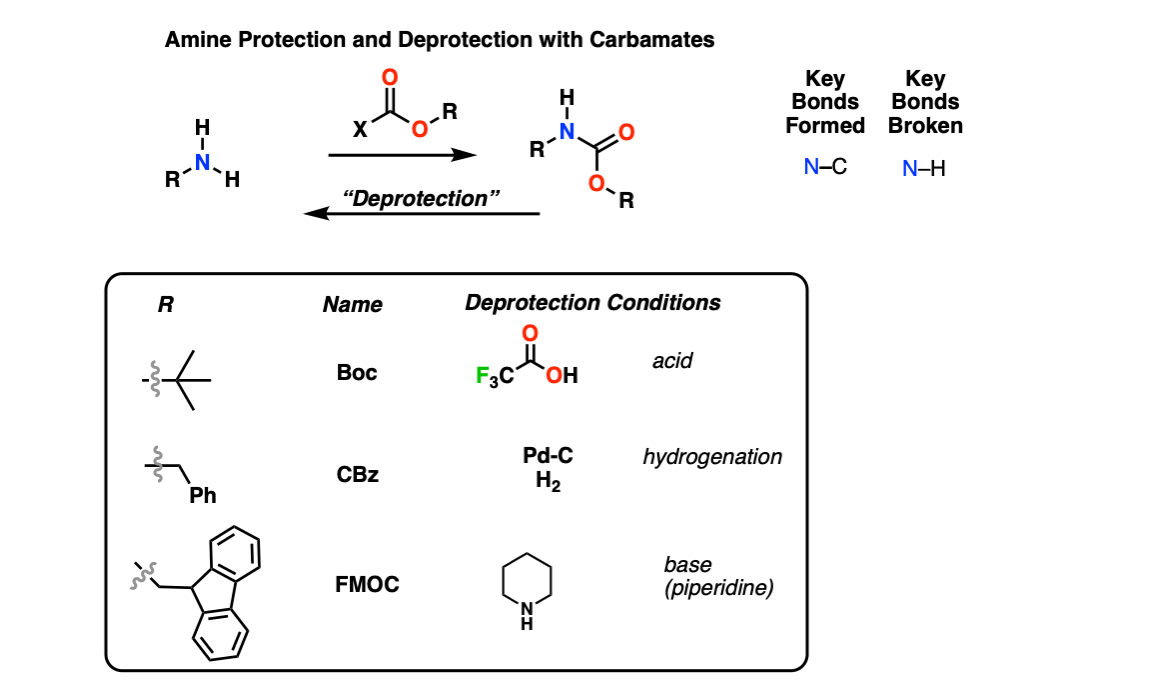
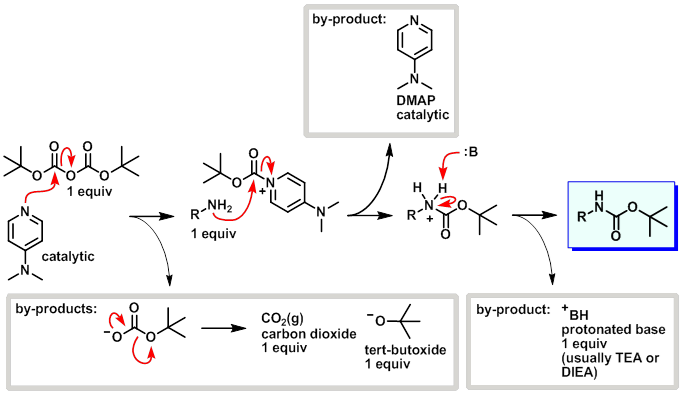
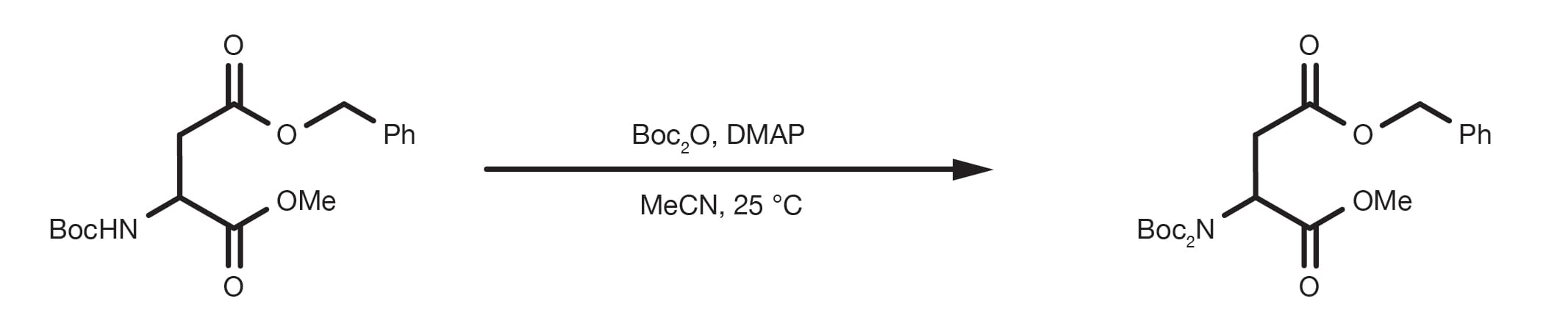
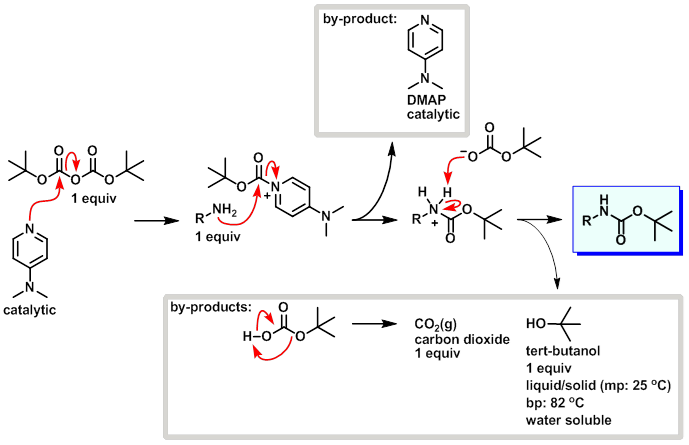
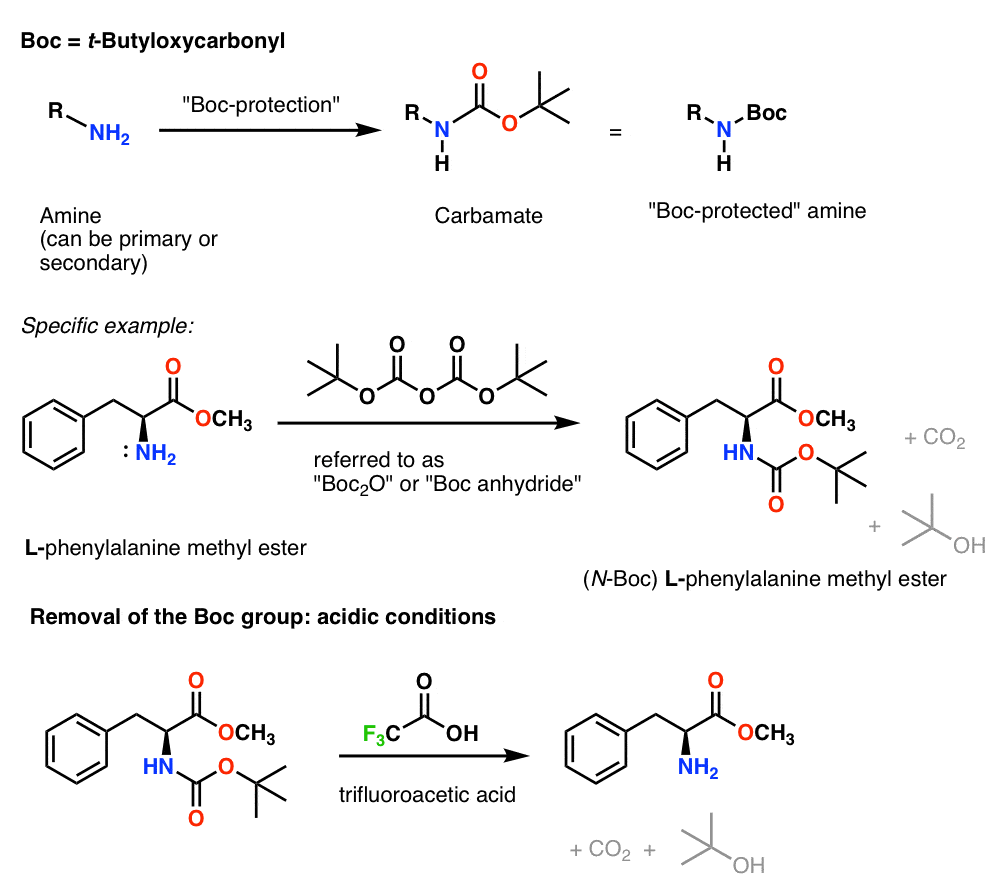
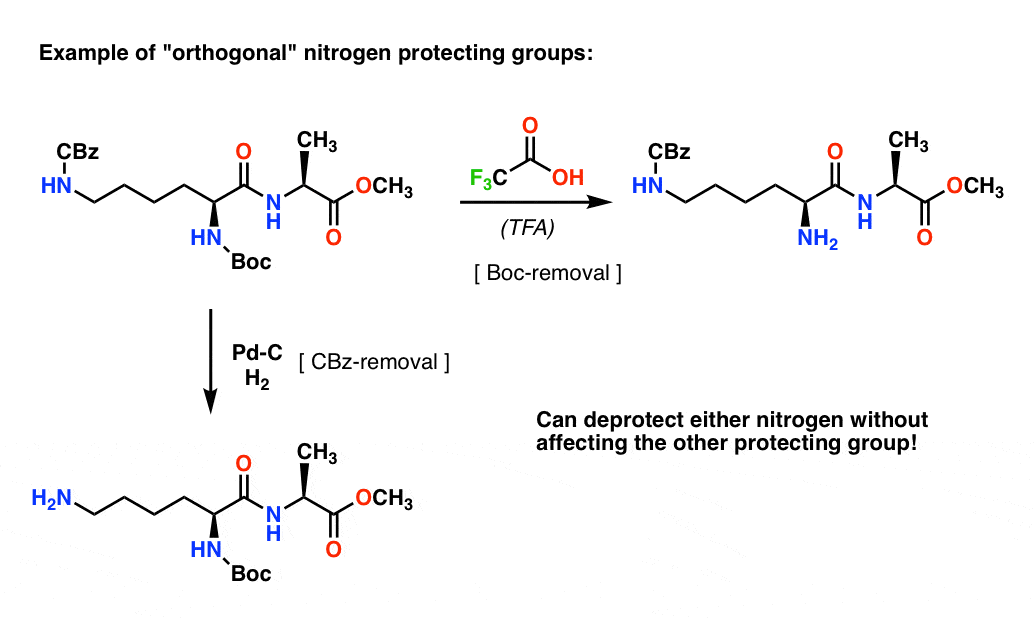
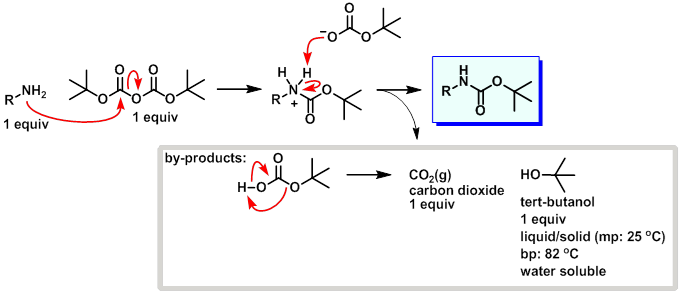
Deprotection of N‐tert‐Butoxycarbonyl (Boc) Protected Functionalized Heteroarenes via Addition–Elimination with 3‐Methoxypropylamine - Gulledge - 2020 - European Journal of Organic Chemistry - Wiley Online Library
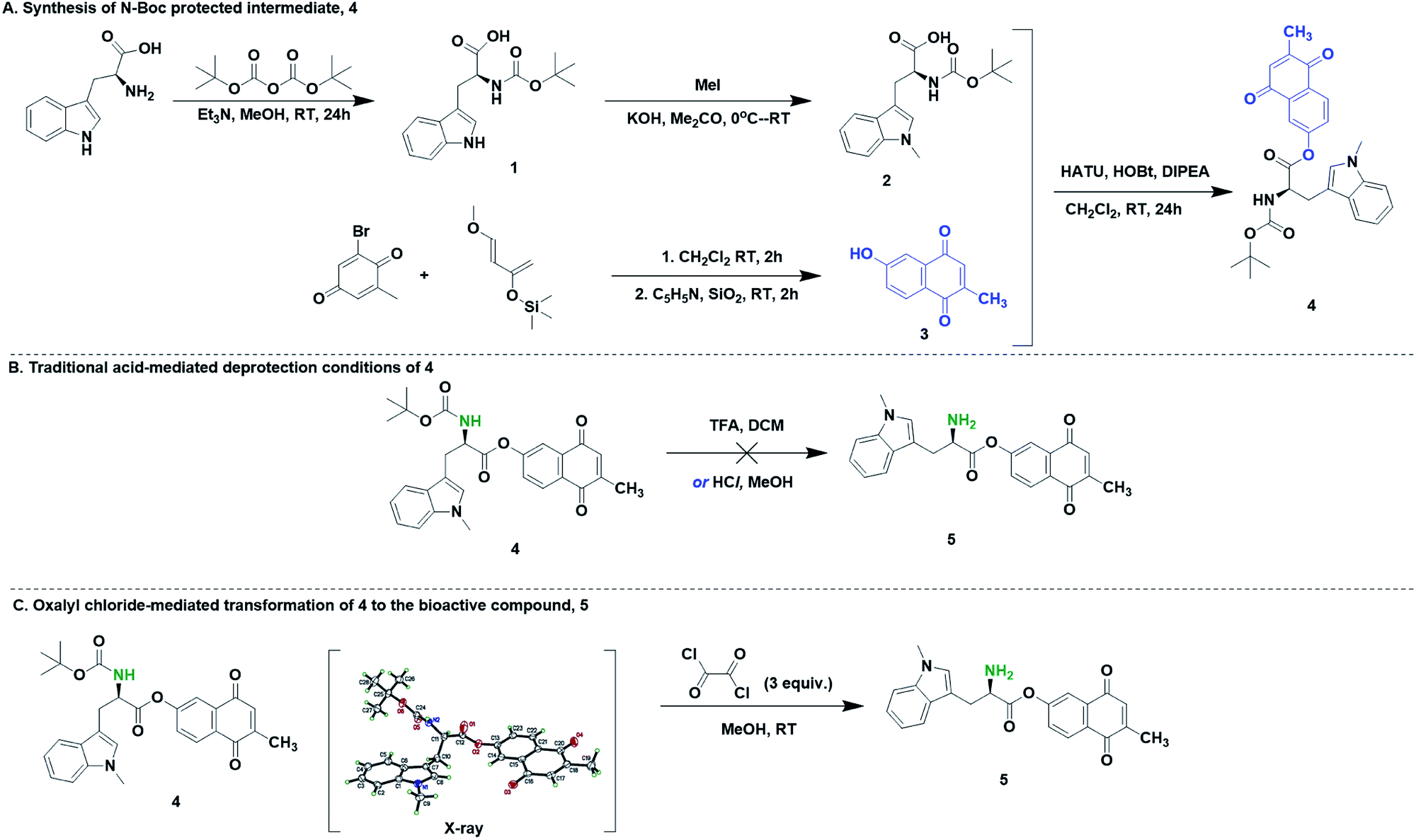
Mild deprotection of the N-tert -butyloxycarbonyl ( N -Boc) group using oxalyl chloride - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04110F

Mixed metal MgO–ZrO2 nanoparticle‐catalyzed O‐tert‐Boc protection of alcohols and phenols under solvent‐free conditions - Gawande - 2012 - Applied Organometallic Chemistry - Wiley Online Library