Novel Formaldehyde-Induced Modifications of Lysine Residue Pairs in Peptides and Proteins: Identification and Relevance to Vaccine Development | Molecular Pharmaceutics
In situ vaccine application of inactivated CPMV nanoparticles for cancer immunotherapy - Materials Advances (RSC Publishing) DOI:10.1039/D0MA00752H

Inactivated or damaged? Comparing the effect of inactivation methods on influenza virions to optimize vaccine production - ScienceDirect

Reactions of β-Propiolactone with Nucleobase Analogues, Nucleosides, and Peptides - Journal of Biological Chemistry
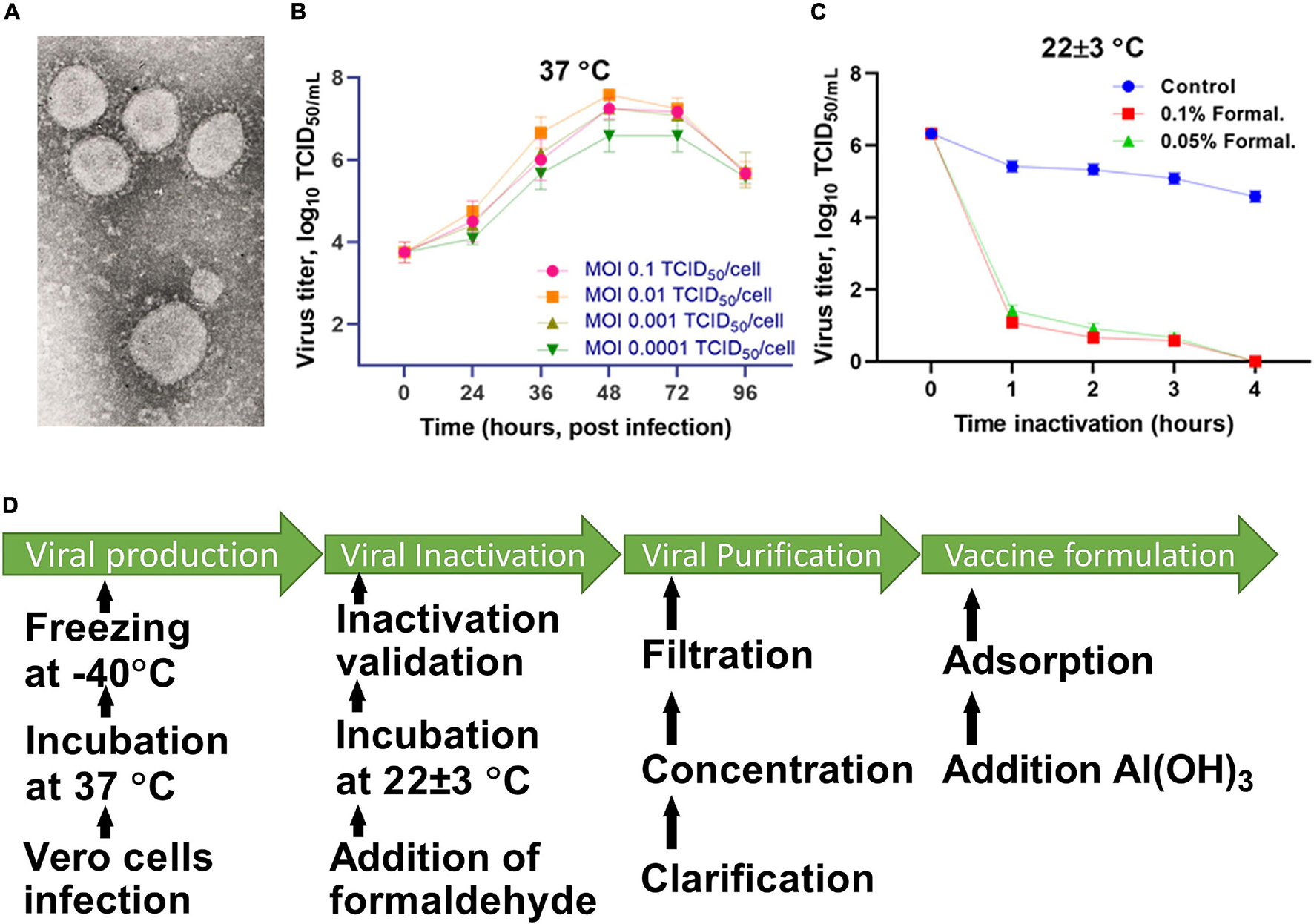
Frontiers | Development of the Inactivated QazCovid-in Vaccine: Protective Efficacy of the Vaccine in Syrian Hamsters | Microbiology
Formalin Inactivation of Japanese Encephalitis Virus Vaccine Alters the Antigenicity and Immunogenicity of a Neutralization Epitope in Envelope Protein Domain III

Inactivation methods for whole influenza vaccine production - Sabbaghi - 2019 - Reviews in Medical Virology - Wiley Online Library
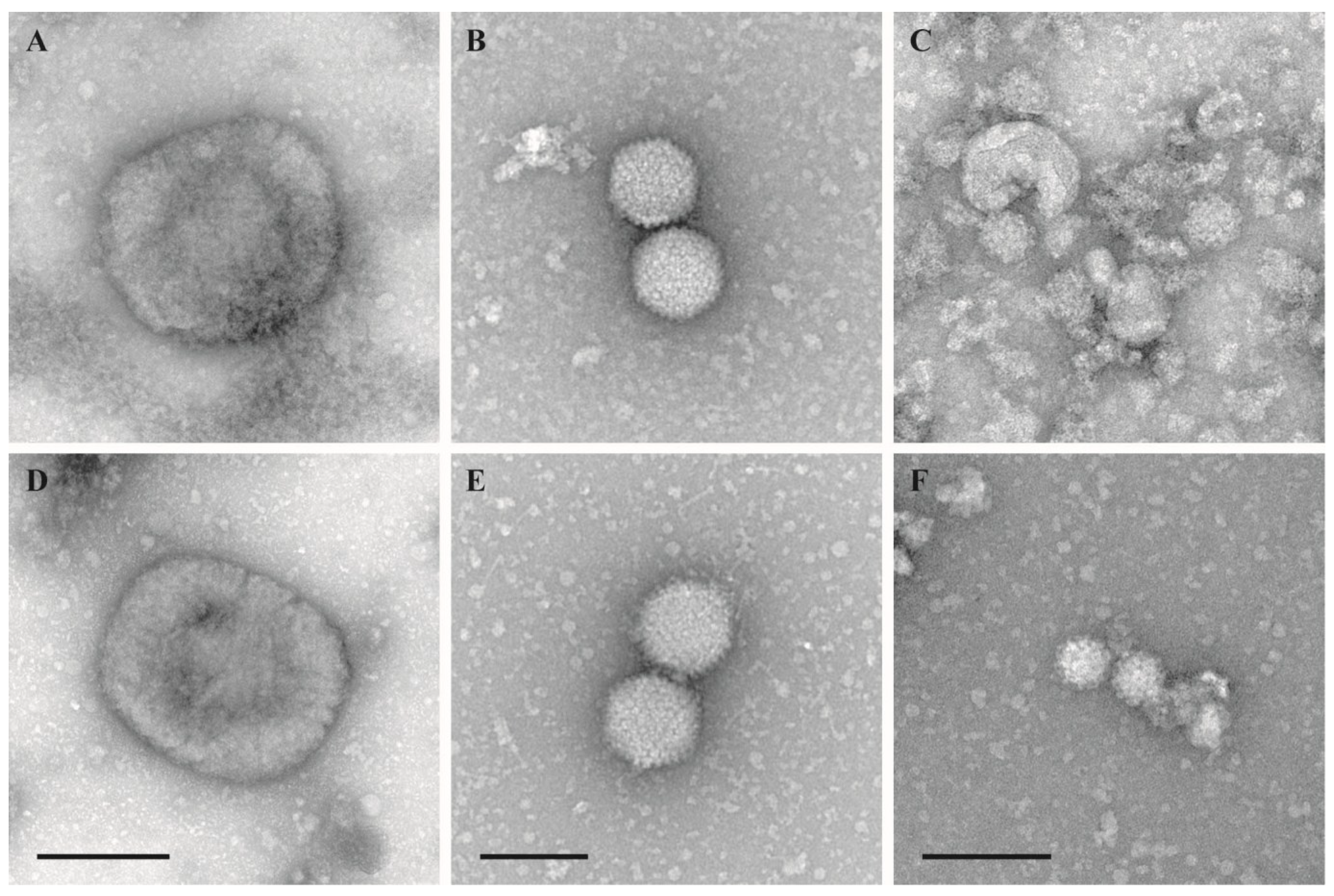
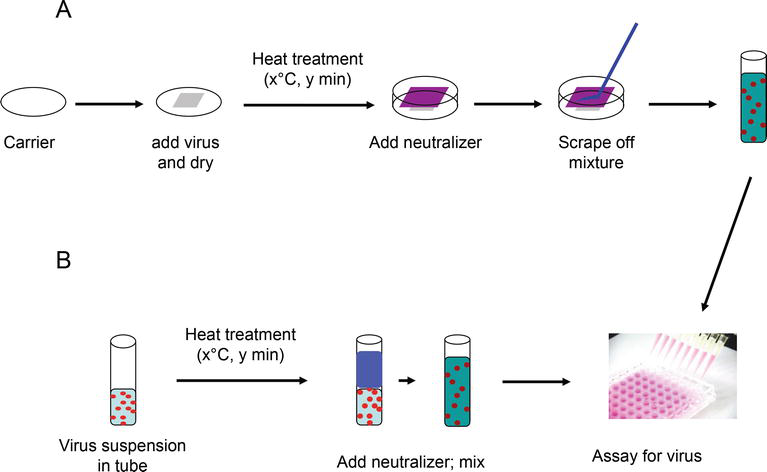
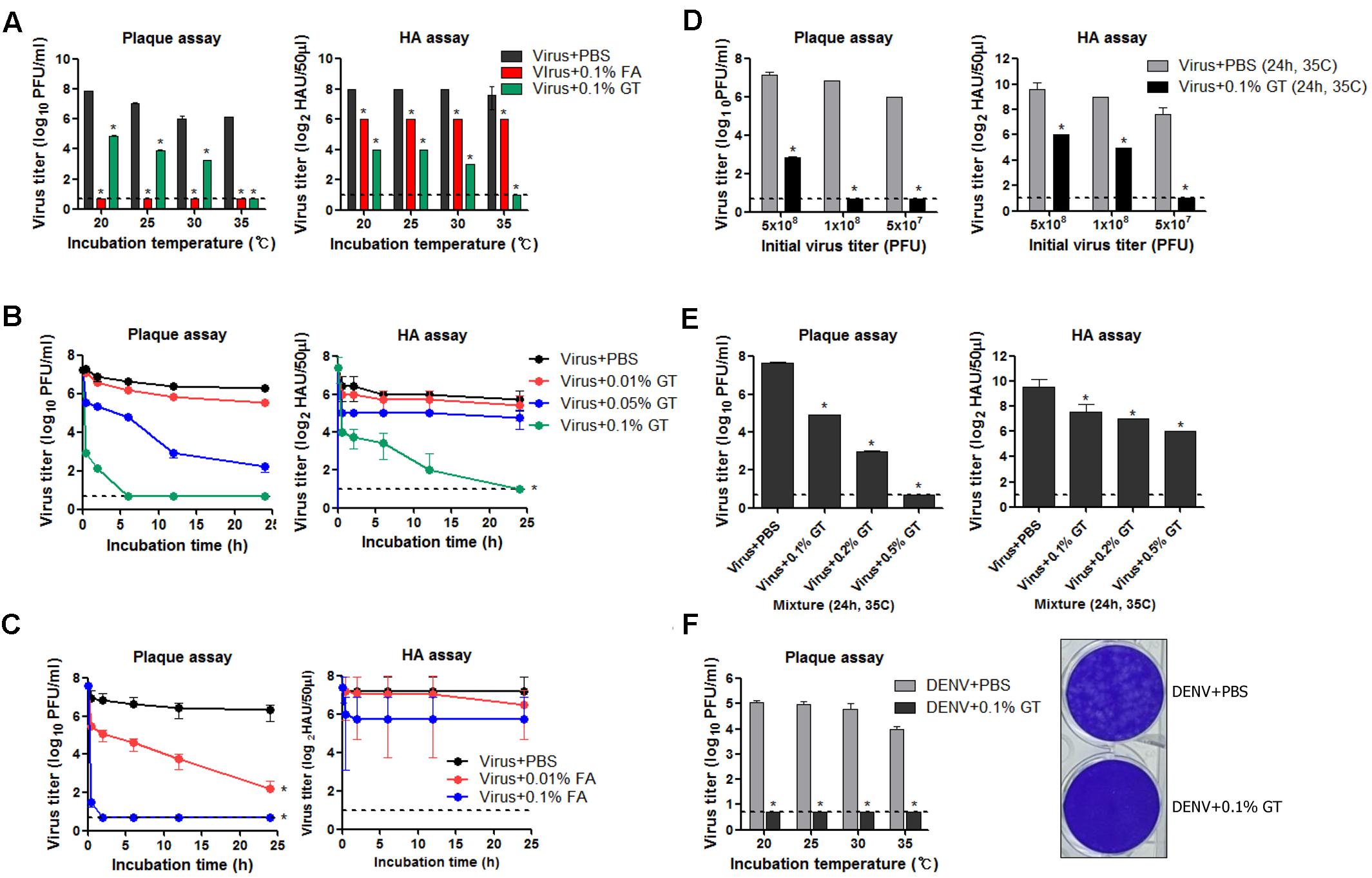
Viruses | Free Full-Text | Evaluation of Virus Inactivation by Formaldehyde to Enhance Biosafety of Diagnostic Electron Microscopy
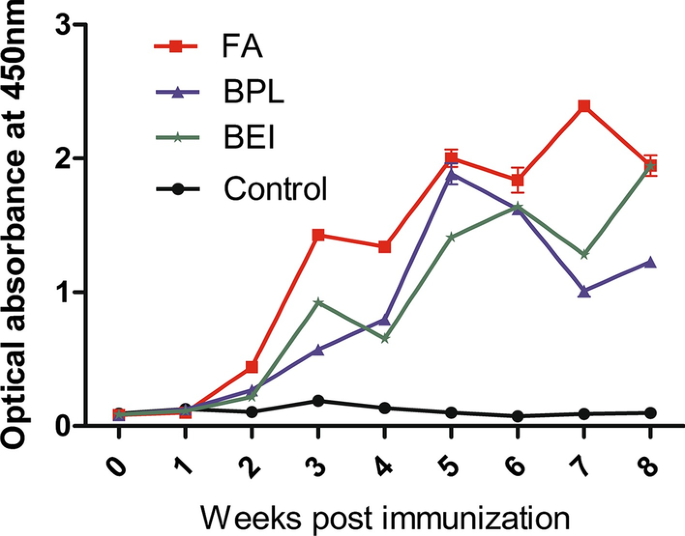
Assessments of different inactivating reagents in formulating transmissible gastroenteritis virus vaccine | Virology Journal | Full Text

Chemical-free inactivated whole influenza virus vaccine prepared by ultrashort pulsed laser treatment

Immunological evaluation of an inactivated SARS-CoV-2 vaccine in rhesus macaques: Molecular Therapy - Methods & Clinical Development

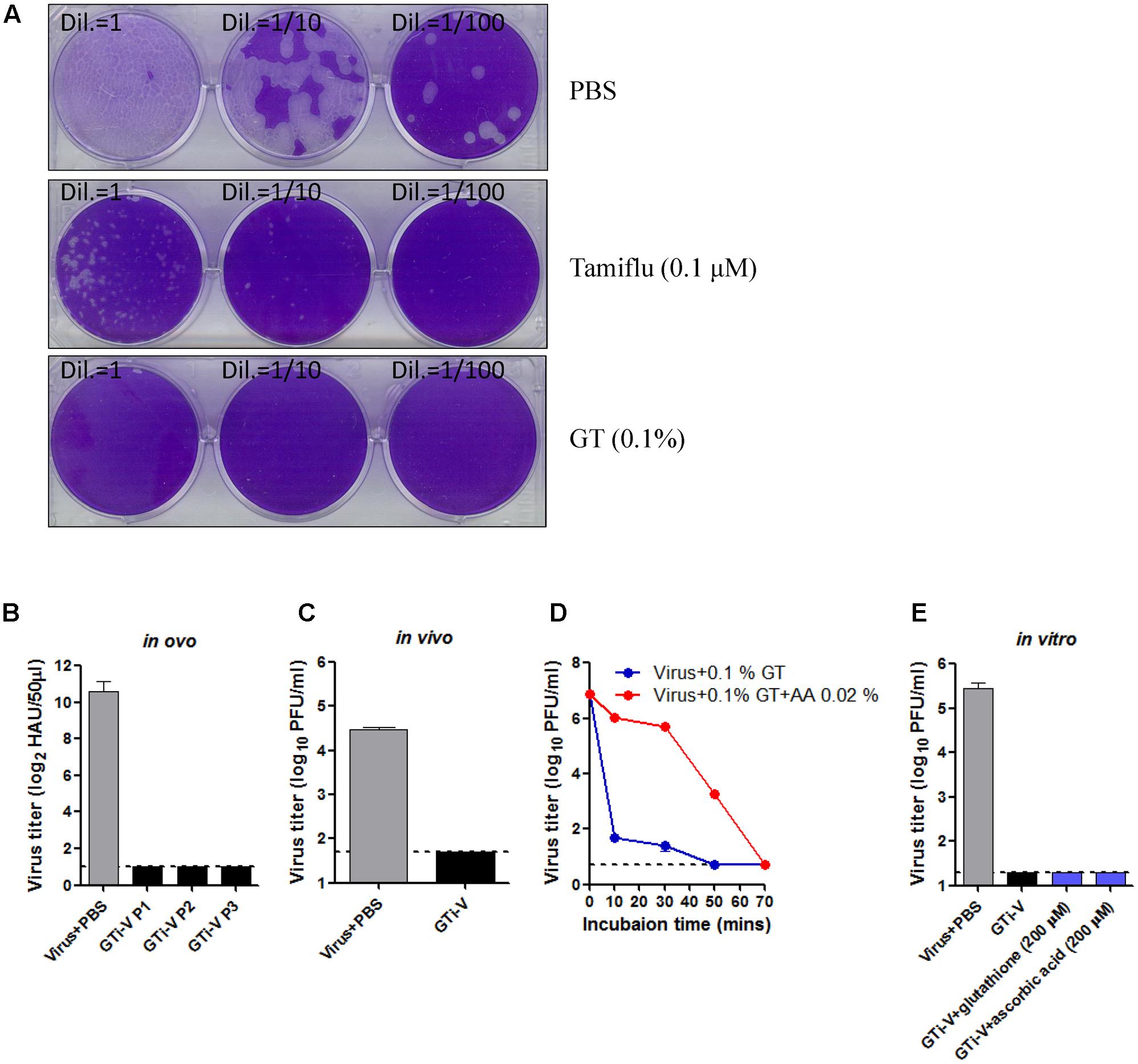
Sanitizing agents for virus inactivation and disinfection - Lin - 2020 - VIEW - Wiley Online Library

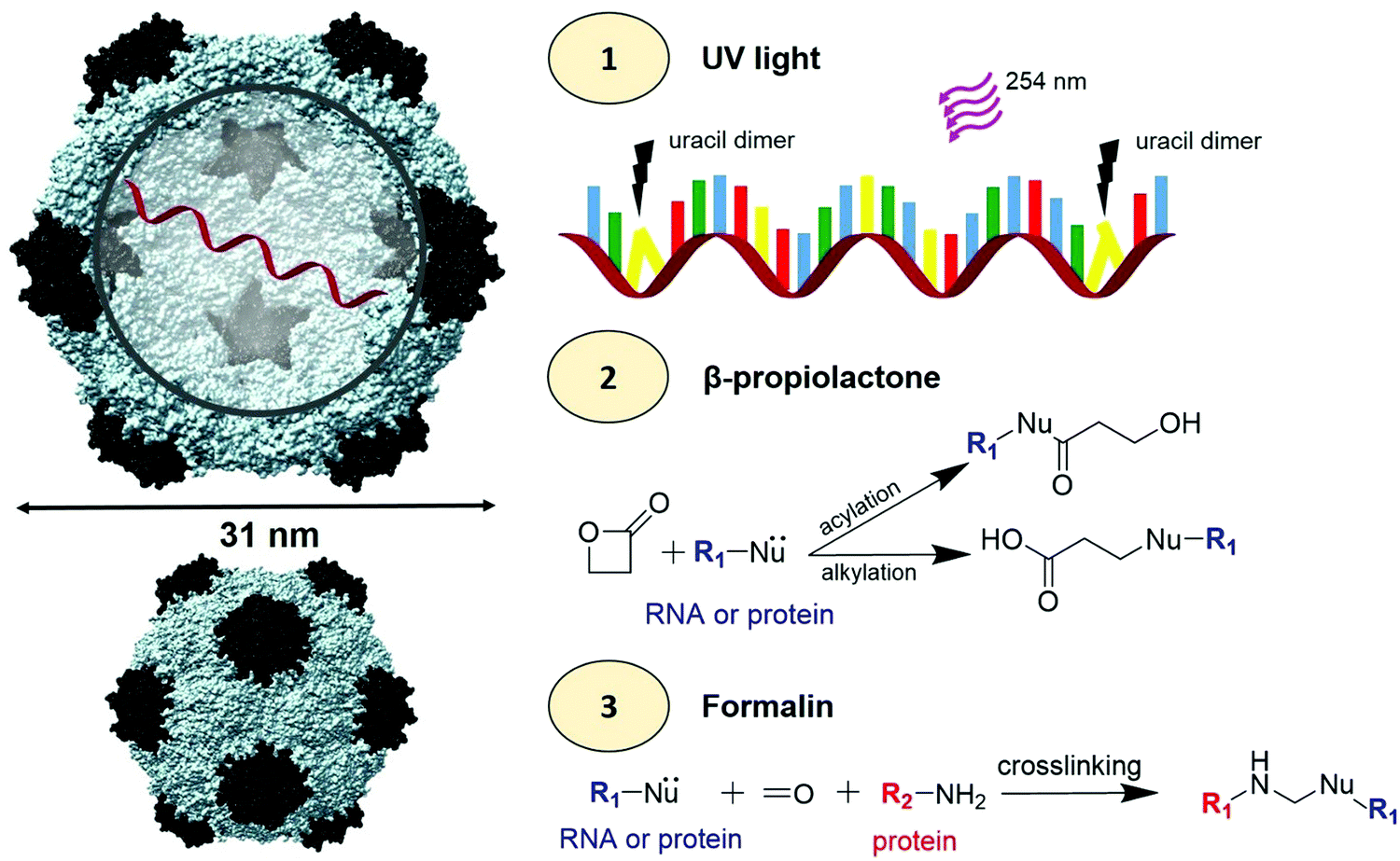
Reaction mechanism of formaldehyde with either DNA/RNA or amino acids.... | Download Scientific Diagram

Sanitizing agents for virus inactivation and disinfection - Lin - 2020 - VIEW - Wiley Online Library
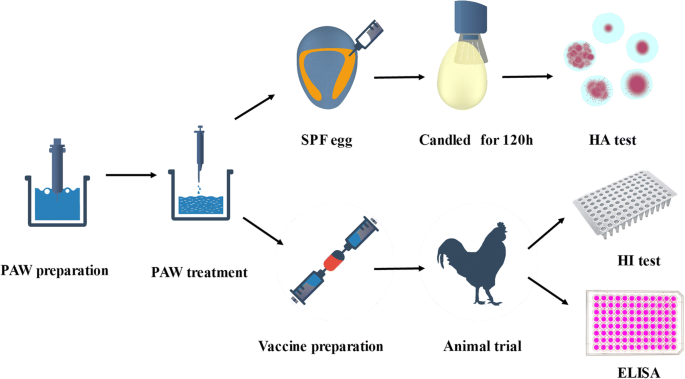
Preparation of the inactivated Newcastle disease vaccine by plasma activated water and evaluation of its protection efficacy | SpringerLink

Purified Inactivated Zika Vaccine Candidates Afford Protection against Lethal Challenge in Mice | Scientific Reports

A comparative study of the effect of UV and formalin inactivation on the stability and immunogenicity of a Coxsackievirus B1 vaccine - ScienceDirect