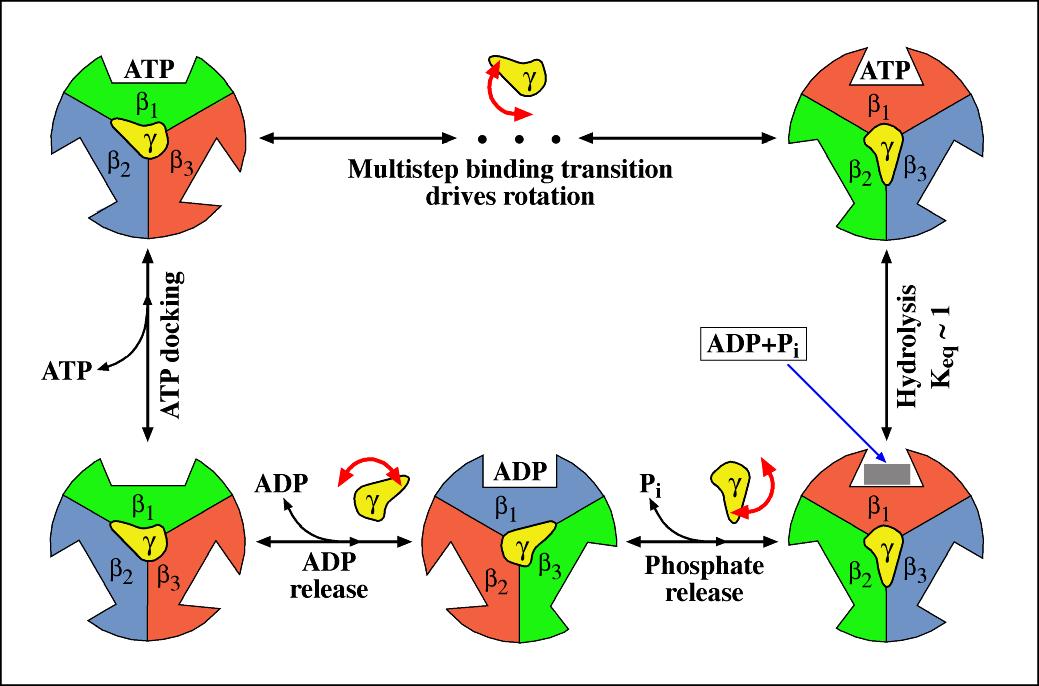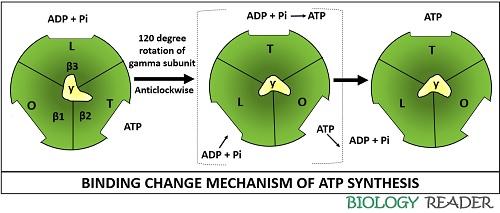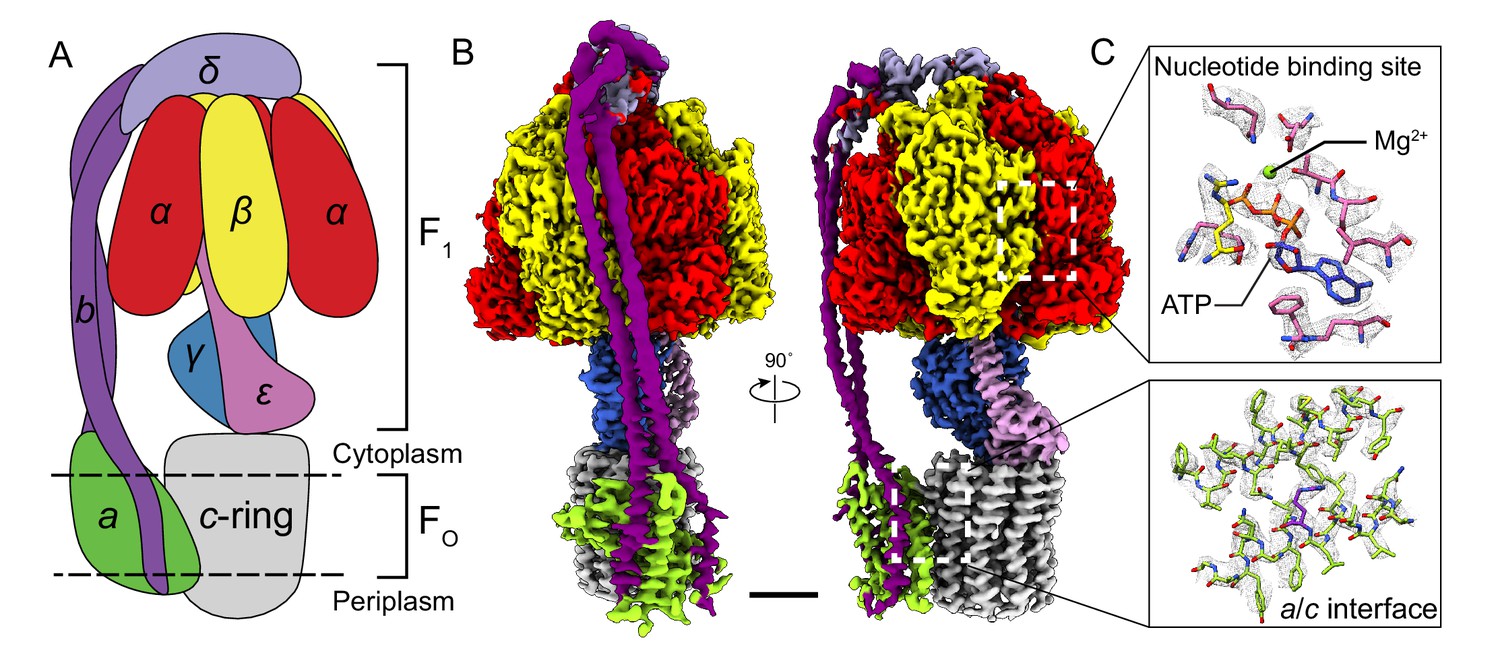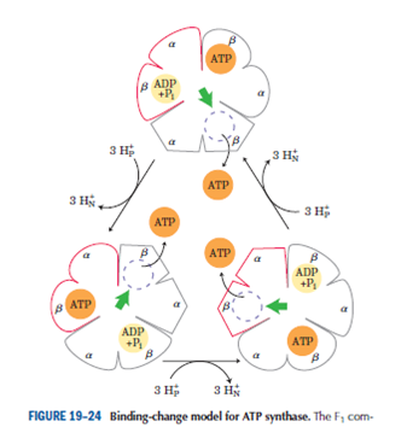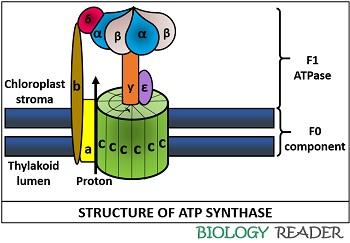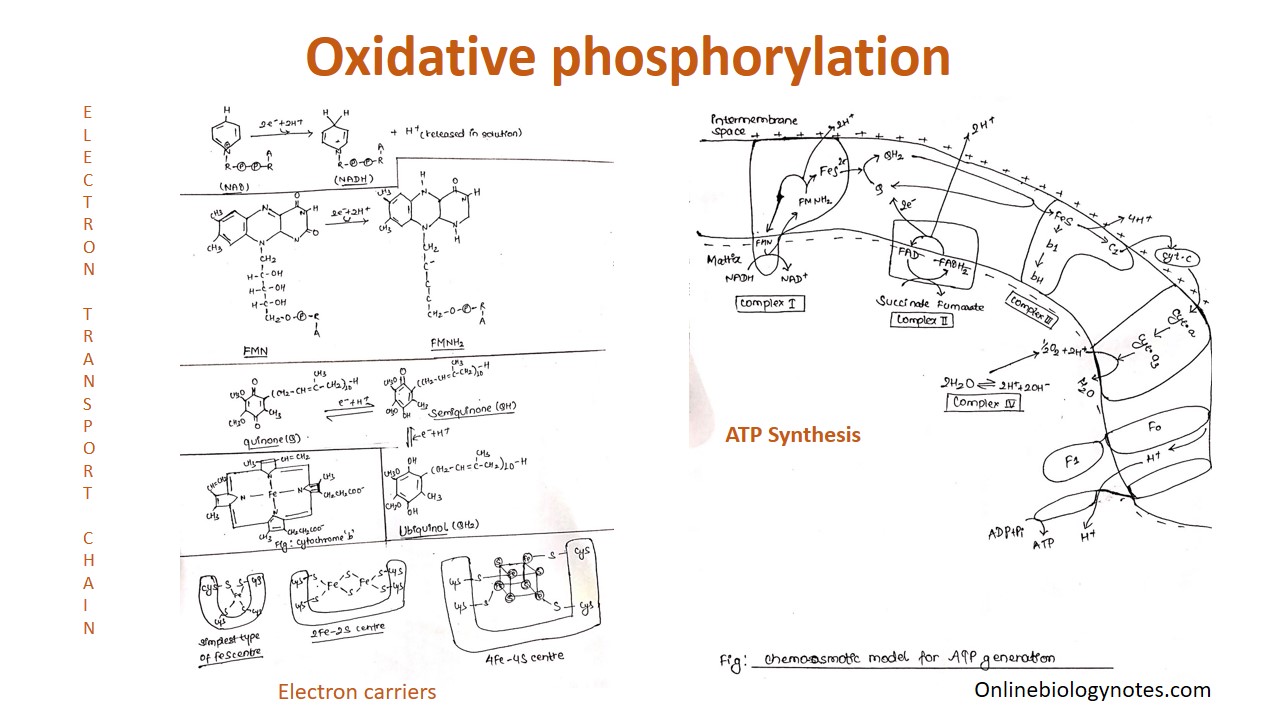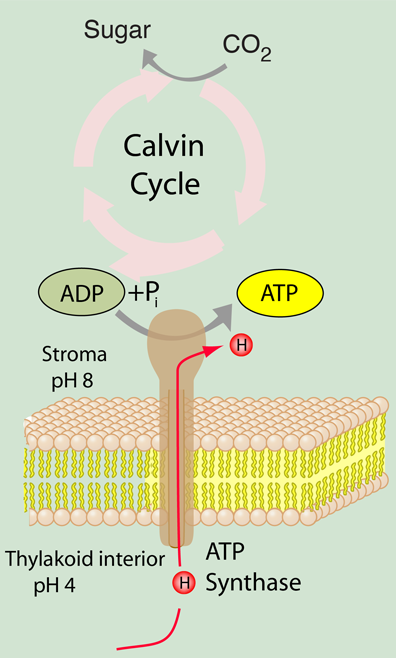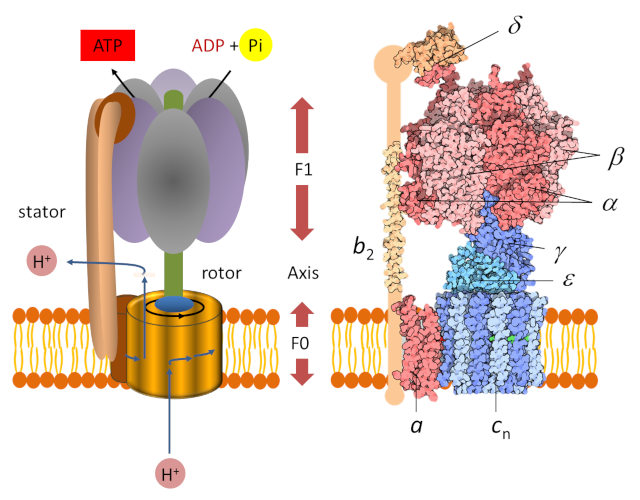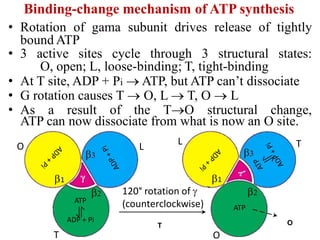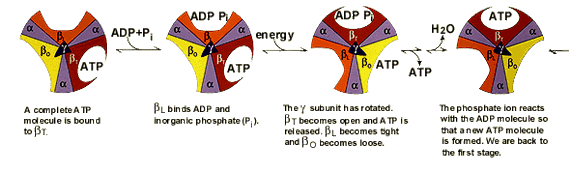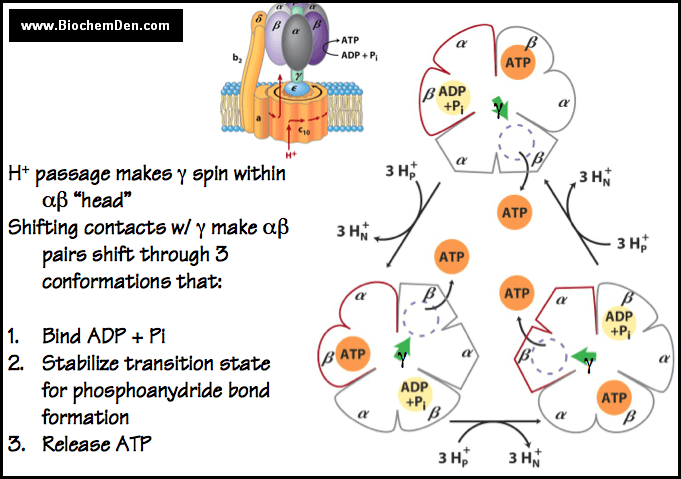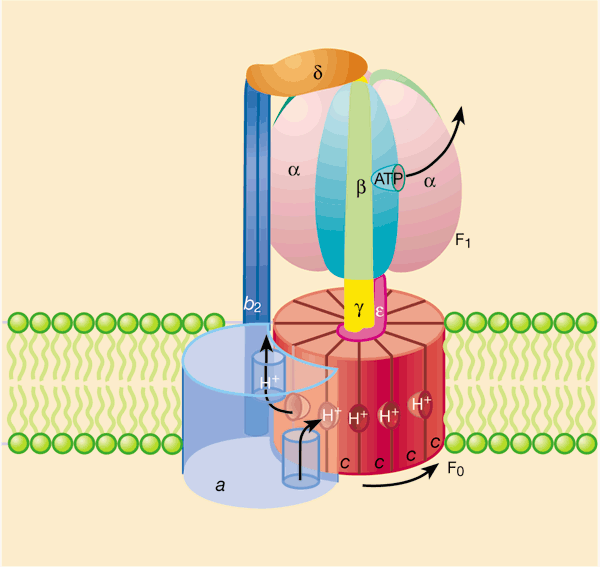
Frontiers | A Review of the Inhibition of the Mitochondrial ATP Synthase by IF1 in vivo: Reprogramming Energy Metabolism and Inducing Mitohormesis | Physiology

Rapid hydrolysis of ATP by mitochondrial F1-ATPase correlates with the filling of the second of three catalytic sites | PNAS

Mitochondrial ATP Synthase: CRYSTAL STRUCTURE OF THE CATALYTIC F1 UNIT IN A VANADATE-INDUCED TRANSITION-LIKE STATE AND IMPLICATIONS FOR MECHANISM - ScienceDirect