Trimethylsilyldiazomethane as a Versatile Stitching Agent for the Introduction of Aziridines into Functionalized Organic Molecules | Organic Letters

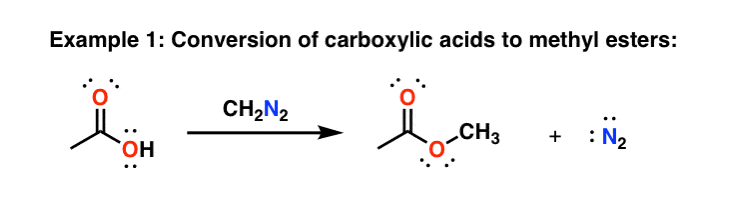
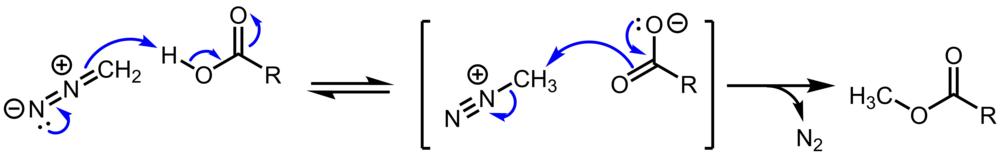
Mechanism of Methyl Esterification of Carboxylic Acids by Trimethylsilyldiazomethane - Kühnel - 2007 - Angewandte Chemie - Wiley Online Library

The reaction of PtdIns(3,4,5)P3 with TMS-diazomethane. TMS-diazomethane... | Download Scientific Diagram

Figure 8 from Methyl esterification of fatty acids and eicosanoids with a novel reagent trimethylsilyldiazomethane for analysis by gas chromatography-mass spectrometry | Semantic Scholar
![Organics | Free Full-Text | Understanding the Reactivity of Trimethylsilyldiazoalkanes Participating in [3+2] Cycloaddition Reactions towards Diethylfumarate with a Molecular Electron Density Theory Perspective | HTML Organics | Free Full-Text | Understanding the Reactivity of Trimethylsilyldiazoalkanes Participating in [3+2] Cycloaddition Reactions towards Diethylfumarate with a Molecular Electron Density Theory Perspective | HTML](https://www.mdpi.com/organics/organics-01-00002/article_deploy/html/images/organics-01-00002-sch003.png)
Organics | Free Full-Text | Understanding the Reactivity of Trimethylsilyldiazoalkanes Participating in [3+2] Cycloaddition Reactions towards Diethylfumarate with a Molecular Electron Density Theory Perspective | HTML

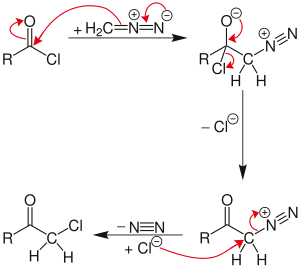
Catalytic and Regioselective Ring Expansion of Arylcyclobutanones with Trimethylsilyldiazomethane. Ligand-Dependent Entry to β-Ketosilane or Enolsilane Adducts | Organic Letters

Mechanism of Methyl Esterification of Carboxylic Acids by Trimethylsilyldiazomethane - Kühnel - 2007 - Angewandte Chemie - Wiley Online Library

Mechanism of Methyl Esterification of Carboxylic Acids by Trimethylsilyldiazomethane - Kühnel - 2007 - Angewandte Chemie International Edition - Wiley Online Library
![Organics | Free Full-Text | Understanding the Reactivity of Trimethylsilyldiazoalkanes Participating in [3+2] Cycloaddition Reactions towards Diethylfumarate with a Molecular Electron Density Theory Perspective | HTML Organics | Free Full-Text | Understanding the Reactivity of Trimethylsilyldiazoalkanes Participating in [3+2] Cycloaddition Reactions towards Diethylfumarate with a Molecular Electron Density Theory Perspective | HTML](https://www.mdpi.com/organics/organics-01-00002/article_deploy/html/images/organics-01-00002-sch004.png)
Organics | Free Full-Text | Understanding the Reactivity of Trimethylsilyldiazoalkanes Participating in [3+2] Cycloaddition Reactions towards Diethylfumarate with a Molecular Electron Density Theory Perspective | HTML

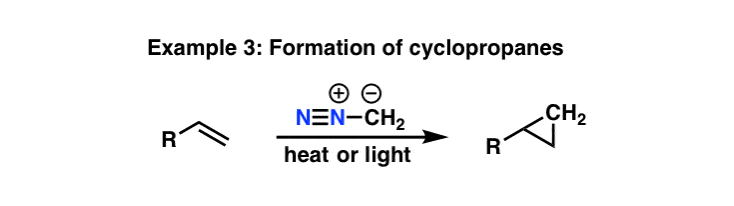
1,3-dipolar cycloadditions of trimethylsilyldiazomethane revisited: steric demand of the dipolarophile and the influence on product distribution. - Abstract - Europe PMC

Reaction mechanism of the methylation of a carboxylic acid R-COOH with... | Download Scientific Diagram